

Lumen Bioscience, Seattle, WA, USA
www.lumen.bio
Stopping a global killer, Lumen Bioscience takes aim at deadly diarrheal diseases in resource-limited settings
Lumen’s low-cost, oral antibody-based cocktail targets key bacterial pathogens
By Jake Siegel
Diarrheal diseases kill 800,000 children each year—more than malaria, measles and AIDS combined.
Lumen Bioscience is taking aim at two of the main culprits: Campylobacter jejuni and enterotoxigenic Escherichia coli (ETEC). With support from CARB-X, Lumen is developing a cocktail of antibody-like proteins designed to bind and neutralize these bacterial pathogens. The intended product is a low-cost, oral preventative that can be easily shipped, stored, and used worldwide, including in regions that lack cold-chain distribution.
“Diarrheal diseases have devastating impacts in the developing world, but the ability to effectively prevent and treat them has been hindered by the global scale of the problem and the extremely high cost of traditional drug development,” says Jim Roberts, M.D., Ph.D., Lumen’s co-founder and Chief Scientific Officer. “Our unique approach offers the potential to provide an effective drug at low cost and massive scale.”
That unique approach is Lumen’s spirulina-based drug development and production platform. The company previously developed novel methods to engineer the widely consumed food algae spirulina to serve as both a low-cost factory and a safe delivery vehicle for antibodies and other protein therapeutics. Moreover, no purification of the therapeutic protein is needed for oral delivery. The final drug product is whole, unrefined spirulina biomass containing large amounts of the therapeutic proteins. The simplicity of downstream processing means that the cost is just pennies per dose, and the robust safety record of both oral therapeutic proteins and spirulina itself reduces the cost and risk of clinical development.
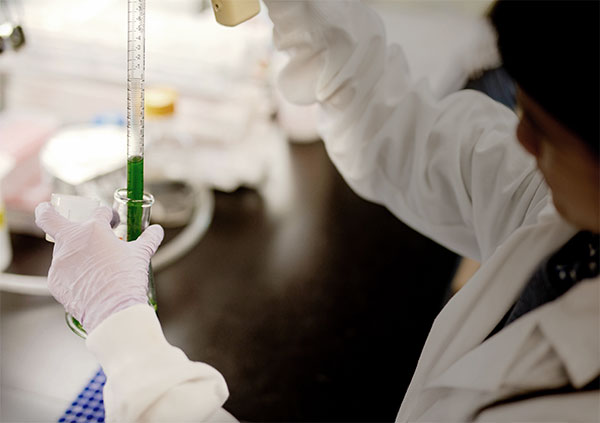
A Lumen scientist works with a prototype antibody-spirulina strain. Photo courtesy of Arianna Lewellyn (Lumen)
Lumen’s therapeutics are derived in part from the antigen-binding domains of camelid single chain antibodies, also called VHHs or nanobodies. Lumen has developed unique protein designs that result in VHHs that possess binding affinities and specificities comparable to IgG antibodies. Dried spirulina biomass protects the VHHs from the harsh gastric environment before delivering them to the gut lumen, where they bind directly to their pathogen targets and block interaction with the host.
Lumen’s scalable drug-making platform opens the door to treating widespread diseases with high complexity VHH cocktails. This is critical for the development of therapeutics that can address the large natural diversity of many important pathogens. The company’s CARB-X project will combine 12-20 VHHs, each attaching to a different target. For both Campylobacter and ETEC, they target key virulence factors: flagellin for Campylobacter and adhesins for ETEC. The goal is to cover—in a single capsule—85% of the diverse bacterial strains that cause human disease.
Lumen’s CARB-X project could help support UNICEF and the World Health Organization’s ambitious goal of ending two major preventable causes of child death–pneumonia and diarrhea–by 2025. Safe, affordable and effective preventative strategies will be key to reaching the most vulnerable populations in low- and middle-income countries. Furthermore, both ETEC and Campylobacter rank high in the US Centers for Disease Control and Prevention (CDC) Antibiotic Resistance Threats Report and WHO priority pathogens list, reflecting the declining ability of existing drugs to cure these infections.
“The development of an oral antibody cocktail that is cheap, scalable, easily administered, and antibiotic-free would represent a significant advance for the prevention of diarrheal diseases,” says Erin Duffy, Ph.D., R&D Chief of CARB-X.
CARB-X has awarded Lumen an initial US$5.35 million to develop its immunotherapy, and the project could be eligible for up to $8.26 million more as project milestones are met. The Lumen work is proceeding in collaboration with the US Naval Medical Research Center, led by Frederic Poly; Tufts University in Boston, led by Charles Shoemaker; the University of Virginia, led by Jim Nataro; and A-Alpha Bio, Inc., a Seattle-based biotechnology company.

Jim Roberts (left) is Lumen’s co-founder and Chief Scientific Officer. Brian Finrow (right) is Lumen’s co-founder and Chief Executive Officer. Photo courtesy of Lumen
Jim Roberts
Co-founder and Chief Scientific Officer
Jim Roberts’s career trajectory aligns precisely with the pioneering breakthroughs in the field of biotechnology. Time spent in the Axel Lab during his student years at Columbia University overlapped with the discovery of gene transfer methods for mammalian cells, and helped lay the groundwork for an involvement with the Fred Hutchinson Cancer Research Center that now spans more than three decades.
Ten years as an Investigator at the prestigious Howard Hughes Medical Institute in the 1990s and early 2000s, followed by six years as Director of Basic Sciences at the Hutch, combined with a career-long involvement in the field of cell cycle and cancer biology to instill a fine-grained grasp of both the importance of a sound conceptual foundation and the disadvantages of a vision that’s too narrowly defined. Of special significance to Lumen, Jim’s front-row seat on the development of the field provides a kind of institutional memory that dates back long before the company’s actual founding.
Brian Finrow
Co-founder and Chief Executive Officer
Brian Finrow brings the skills and experience of a lawyer to a position that, in the biotech world, is more typically occupied by a scientist. This is essential to an enterprise like Lumen, which is rethinking drug development from the ground up to emphasize speed, efficiency and ease of delivery, and whose approach is so innovative that traditional ways of thinking about IP and regulatory pathways oftentimes do not apply.
Prior to co-founding Lumen, Brian oversaw complex negotiations with various major biopharma companies and managed IP strategy for Adaptive Biotechnologies, where he was Senior Vice President and General Counsel. As senior attorney at the law firm Cooley LLC, his practice focused on equity financing and M&A transactions, and negotiating complex biotech licensing and collaboration deals. With over 15 years of legal and commercial experience at these market-leading, innovative firms, he is well positioned to develop creative, win-win deal structures with other organizations.
For more information, www.lumen.bio
Published on January 10, 2022


